Enhanced Paveway™ PLUS Selects Expression Organisms Based on Titer Plus Quality
Launched in 2007, our core microbial expression system Paveway™ has offered industry leading titers by enabling lead strain selection based on product titer in six weeks from program initiation, that has resulted in the successful expression of 150+ proteins. Advanced quality analytics are often required to detect differences in protein structure, including post-translational modifications, but these methods typically require enough product that is at least partially purified. The conundrum is that purified product is often not available until a purification process is defined. As such, variations in product quality often are not discovered until purified product is available in quantities sufficient for analysis.
Our enhanced Paveway™ PLUS platform meets these challenges through an enhanced workflow providing exceptional product titers for a wide range of soluble and insoluble proteins, while maintaining extremely tight control of pre-induction product expression. Importantly, fermentations can be run without antibiotic selection pressure while sustaining high plasmid retention, helping avoid regulatory concerns around potential antibiotic resistance and clearance. This new and enhanced workflow combines rapid evaluation of purified product with advanced protein characterization to enable lead strain selection based on both titer and quality in eleven weeks, as the illustrated in Figure 1. With the ability to confirm the identity of both product and key post translational modifications, Paveway PLUS workflow enables early selection of most suitable capture resins for downstream purification, in addition to incorporating modules for specific investigations, such as periplasm and expression studies, and solubilization development.
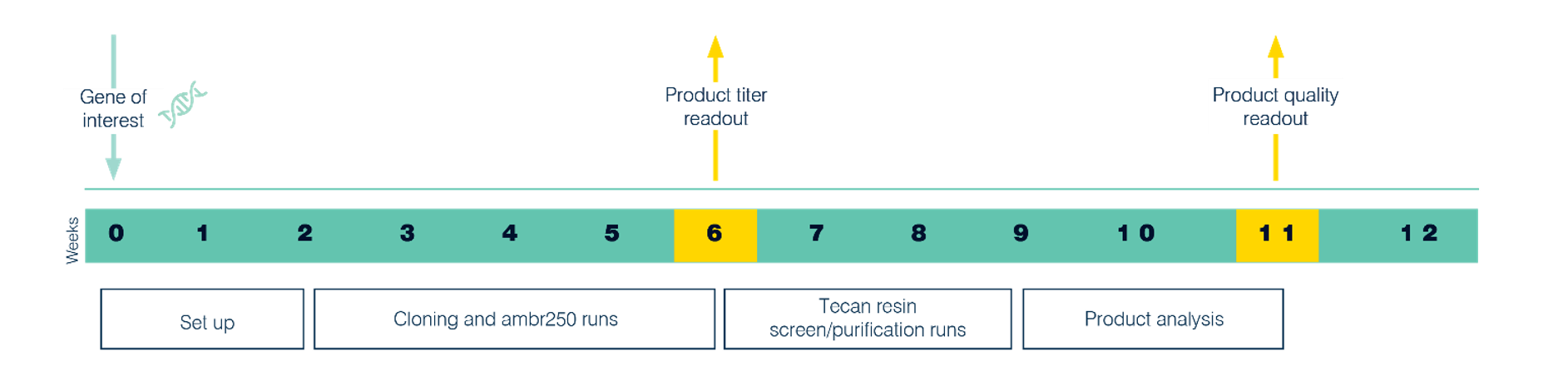
Figure 1. Timeline for Paveway PLUS enhanced workflow.
Paveway PLUS projects begin with cloning a customer-supplied gene into one of our vectors and host strains and applying predefined screens in ambr® 250 microbioreactors, where subsequently samples are prepared for metabolites analysis, cell density and protein titer. This process affords up to eight cultures that once taken through microscale purification produces sufficient material for subsequent high-throughput analysis via UV, SDS PAGE, SEC-HPLC, intact mass spectrometry and isoelectric focusing. As such, in just eleven weeks after beginning the project, strains are selected based on quality as well as productivity.
Case Study: Not by Titer Alone
Designed ankyrin repeat proteins (DARPins) are a promising class of non-immunoglobulin proteins that can offer advantages over antibodies for target binding in drug discovery and drug development. Due to their small size and stability, DARPins are also suited for in vivo imaging or delivery of toxins directly to diseased tissues. DARPins are readily expressed in bacteria and may be multi-functional, for example with one “epitope” targeting an organ, and another with affinity to a specific target within that organ.[1]
Off7, the first engineered DARPin, binds to maltose binding protein with high affinity/specificity. DARPin off7, a prototype for genetically engineered antibody-mimetic protein of interest in drug development, is a 160 amino acid polypeptide with a molecular weight of 17.2 kDa expressed in E. coli. The purpose of this study was to demonstrate the enhanced workflow benefits in producing DARPin off7 in two different strains of E. coli, one of which is known to incorporate undesired gluconic acid post-translational modifications.
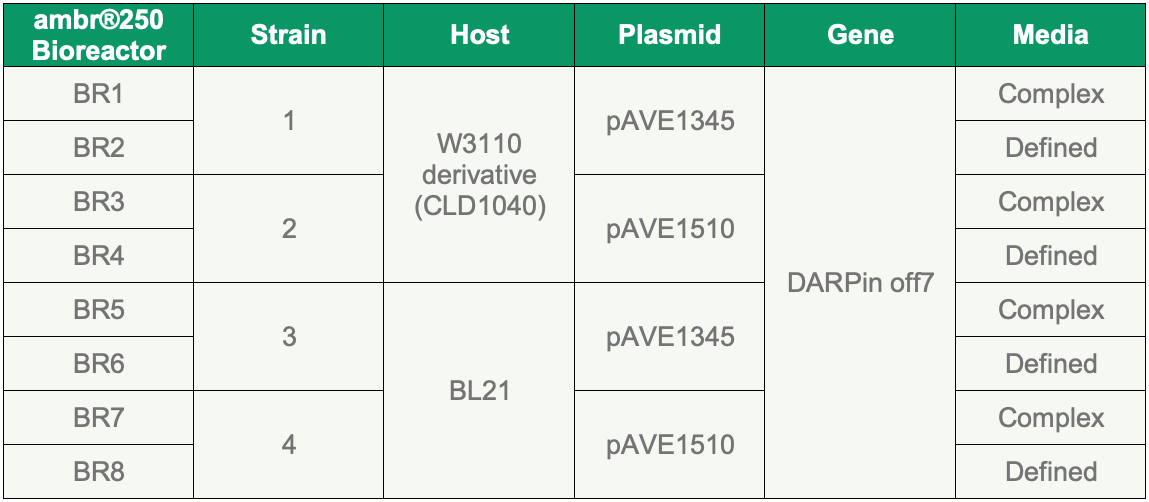
The study was conducted in eight ambr® 250 bioreactors split by two expression hosts: CLD1040, a FUJIFILM Diosynth Biotechnologies strain, and BL21, a common strain of E. coli used in protein expression. Genes were cloned into two different expression vectors (pAVE1345 and pAVE1510), which provide slightly different expression kinetics, and further split by media type (defined or complex) to give eight different sets of conditions as outlined in Table 1.
Table 1. Variable parameters for all replicates.
Each bioreactor (BR1 to BR8) was run under standard control conditions as described below in Table 2.
Table 2. Standard control conditions used for all microbioreactors.
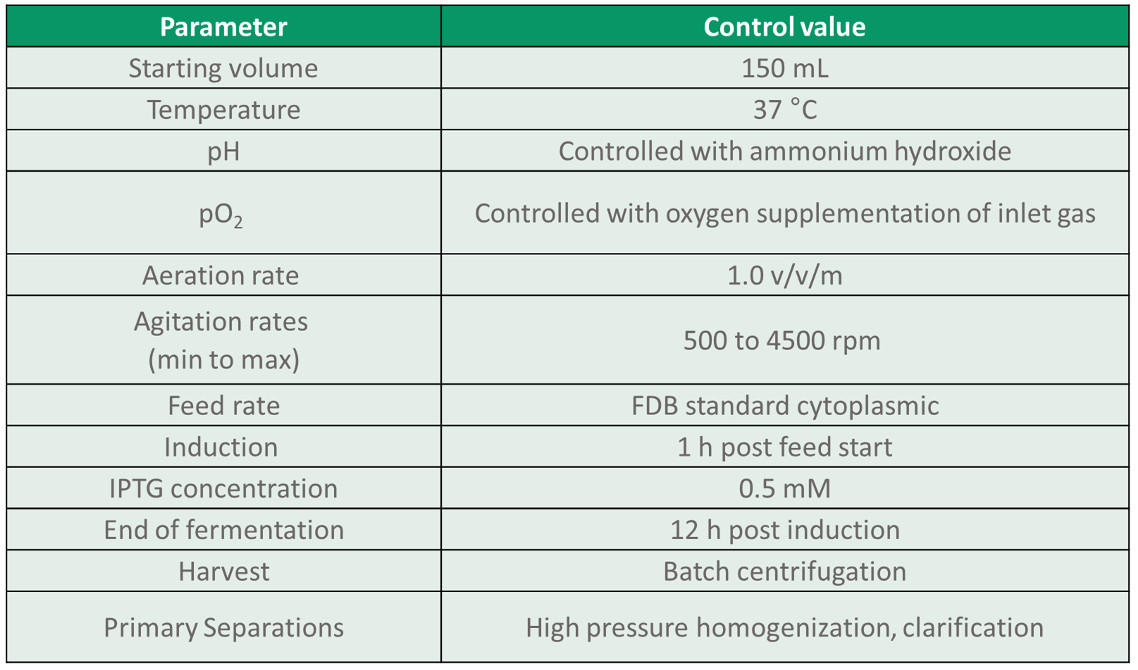
Figure 2 represents process variables monitored during a microbial fermentation: dissolved oxygen (DO) (red line), pH (blue), stir speed (green) and temperature (orange).
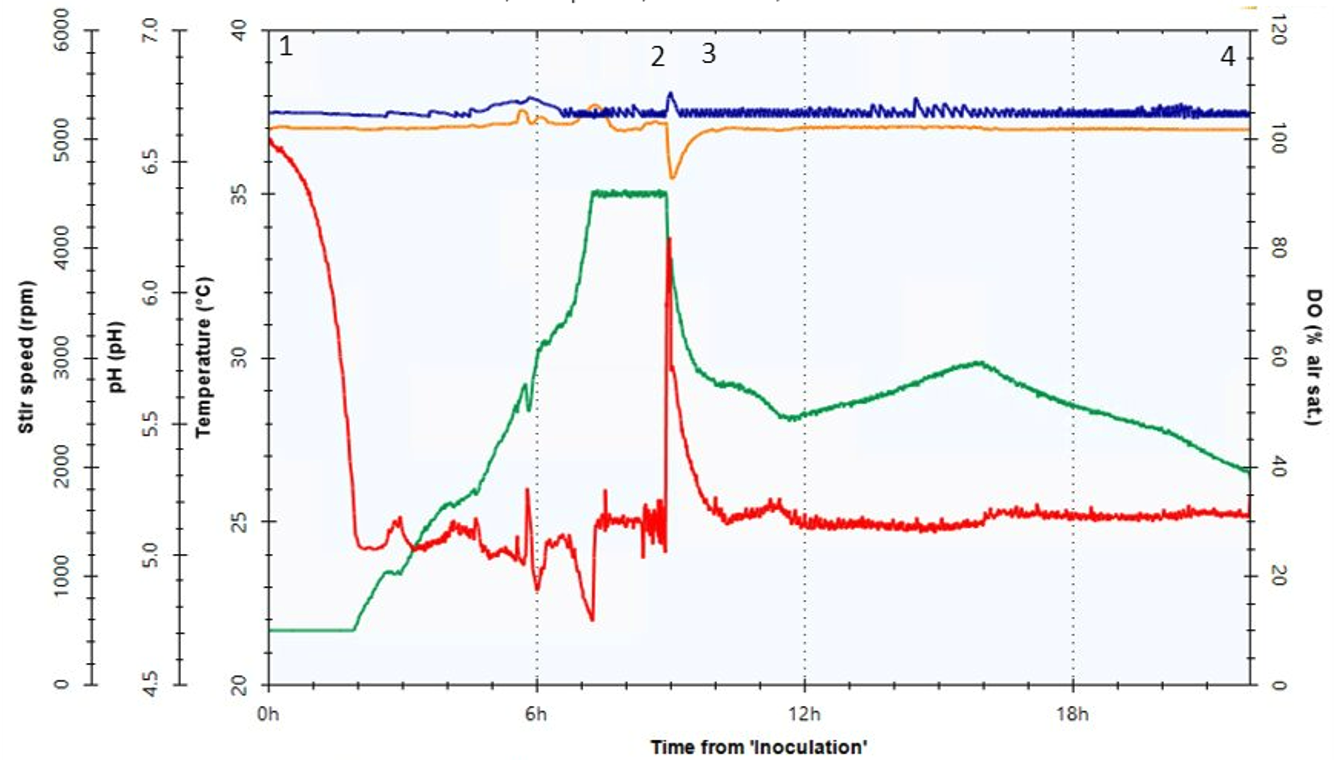
Figure 2. Monitoring of critical process variables over time during a microbioreactor fermentation.
Point 1 indicates bioreactor inoculation and batch phase initiation. The batch phase begins at 100% Dissolved Oxygen (DO) saturation and stirrer speed at zero, DO then falls to the setpoint of 30% and stirrer speed rises to a maximum of about 4000 rpm to maintain this setpoint level. As cells grow, they consume nutrients and oxygen, and by the time they reach point 2 (depletion), they have run out of carbon. At this point, DO levels rise again, which is the signal for the system to initiate feeding. After induction, at point 3, the process is controlled with a capped constant feed rate to maximize protein expression, to the end of fermentation where cells are harvested (point 4).
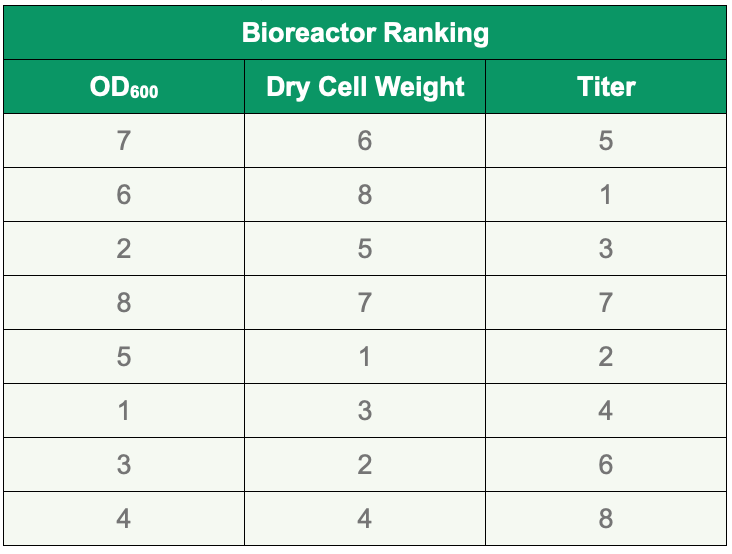
To rank bioreactor performance, growth by cell density (OD600), dry cell weight (in g/L) and titer (g/L) were measured for all eight reactions. Biomass measurements showed a range of OD600 readings ranging from about 100 to 140. Bioreactors BR7, BR6, BR2, and BR8 achieved the highest OD600 readings, while reactions in BR6, BR8, BR5, and BR7 showed the highest dry cell weights. Polyacrylamide gel electrophoresis determined that the four conditions using complex media, bioreactors BR5, BR1, BR3, and BR7, produced the highest titers. These four conditions, selected based on titer were then subjected to the Paveway PLUS enhanced workflow to assess product quality. Table 3 ranks bioreactor runs for OD600, dry cell weight and titer.
Table 3. Ranking of microbioreactor runs according to OD600, dry cell weight and titer.
As the next step in the workflow, material from one of the non-selected bioreactors was then subjected to microscale primary separations to generate sufficient material for high-throughput resin screening using the TECAN Freedom EVO® liquid handling system (to conserve material). The screens were comprised of the following: cation exchange and anion exchange chromatography at four different pH values (8, 7.5, 7, and 6), Capto Q anion exchange resin operated in bind-elute mode, and Capto S cation exchange resin operated in flow-through mode at pH 7, 6, 5, and 4. These resins were selected for their affinity to a broad range of molecules. Under these conditions DARPin binds to Capto Q but not to the cation exchanger. On the basis of product recovery and purity, Capto Q at pH 7.5 was selected for subsequent quality determinations.
As previously described, having determined the optimal binding conditions, the top four producers (BR5, BR1, BR3, and BR7) underwent microscale primary separations and clarification, followed by preparative chromatography using the Cytiva ÄKTA™ Avant 25 system running a HiScreen™ Capto Q™ with a 10-cm bed height and 4.7 mL column volume. Binding buffer used was a phosphate buffer without NaCl at pH 7.5, while the elution buffer contained NaCl. Figure 3 shows chromatograms for the top four producers. Here, to push the limits of the analytics, both wide and narrow product cuts (DARPin off7 pools) were taken to determine the influence of product purity on the analytical method reliability.
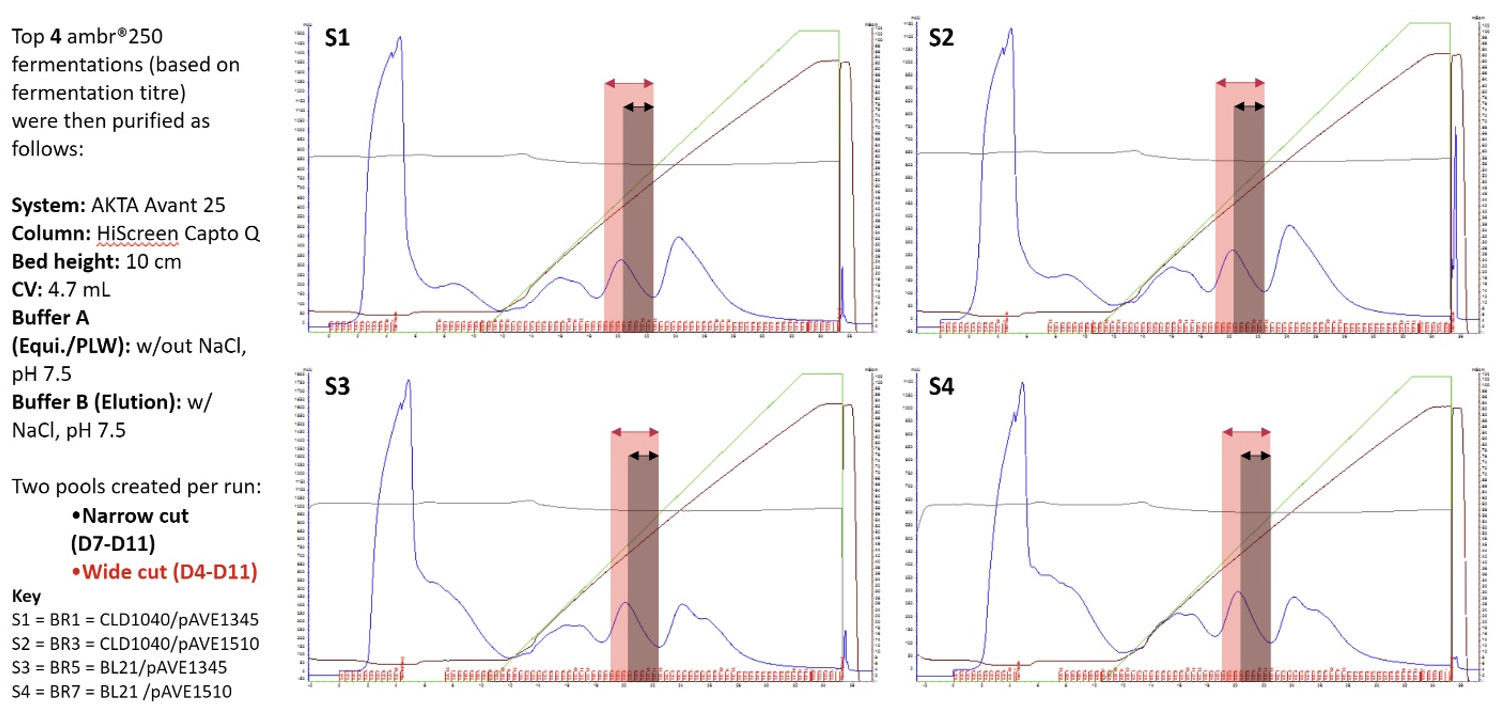
Figure 3. Chromatography runs for the four selected bioreactors. DARPin off7 pools were created for further analysis to determine the influence of product purity: narrow cut (black arrows), wide cut (red arrows).
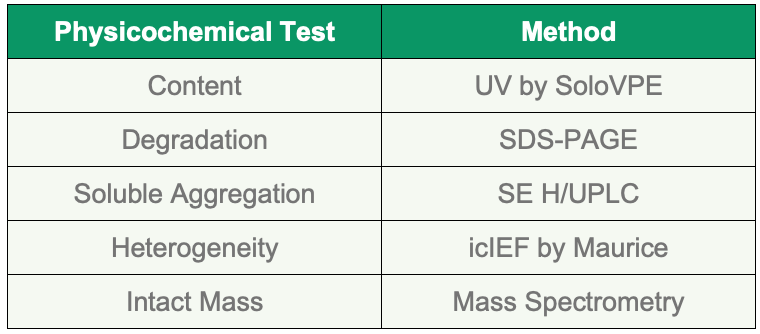
Five methods were used for assessing critical quality attributes for DARPin off7 pools, as listed in Table 4.
Table 4. Quality attributes and methods used.
Analytical methods to determine product quality were applied to both narrow and wide cut samples. As such, the degree of background contaminants varied allowing an evaluation of the sensitivity of each method. The top four producers (both the narrow and wide pools) were then analyzed by size exclusion chromatography to quantify levels of monomeric product or any significant molecular weight excursions due to the presence of truncated DARPin or aggregates. Purity by size exclusion chromatography broadly aligns with observations from SDS-PAGE, which resulted in the 85% to 95% range. As with SDS-PAGE, no significant differences were identified among the four bioreactors, and purity values for the narrow cuts were 2-4% higher than those for the wide cuts.
Analysis of the four candidates through image capillary isoelectric focusing (icIEF) for charge variants also showed minor purity improvements favoring narrow-cut fractions, but this method uncovered the first significant differences between the four lead candidates, and the most striking purity disparities between narrow and wide cuts. DARPin product, with a pI of around 4.3, predominated in every run, but samples 1 (BR1) and 2 (BR 3) showed the highest purity levels and the lowest concentration of post-translationally modified species. Samples 3 and 4 displayed significantly larger peaks with a pI of 4.17, indicating the presence of large amounts of an acidic variant not observed to the same degree in samples 1 and 2. Percentage area of the main peaks from narrow and wide cuts, respectively, improved in sample 1 from 55% to 79%, but for sample 2 the values were nearly identical (85.5% for wide, 84.8% for narrow). For sample 3 improvements of narrow over wide cuts were about 10 percentage points but overall expression of the correct DARPin isoform was less than 60%. For sample 4 both “correct” peaks came in at around 62%. Overall, the observation of a significant acidic variant in S3 and S4 (BL21 strain samples) was noted.
The final method applied to these samples was intact mass using a Waters BioAccord instrument. For narrow-cut samples 1 (BR1) and 2 (BR3), we observed a MW of 17029 Da, corresponding to the theoretical mass peak (17160 Da) minus methionine, and no other peaks of significance. Samples 3 (BR5) and 4 (BR7) produced two peaks that integrated approximately equally. Peak 1 demonstrated a MW of 17160 Da, corresponding to a theoretical mass minus methionine as above. Peak 2, with a MW of 17207 Da, showed both the loss of the methionine as well as the presence of a gluconoyl adduct (Figure 4). It was noted that significant gluconoylation in the BL21 strain samples gave rise to acidic variants detected by icIEF.
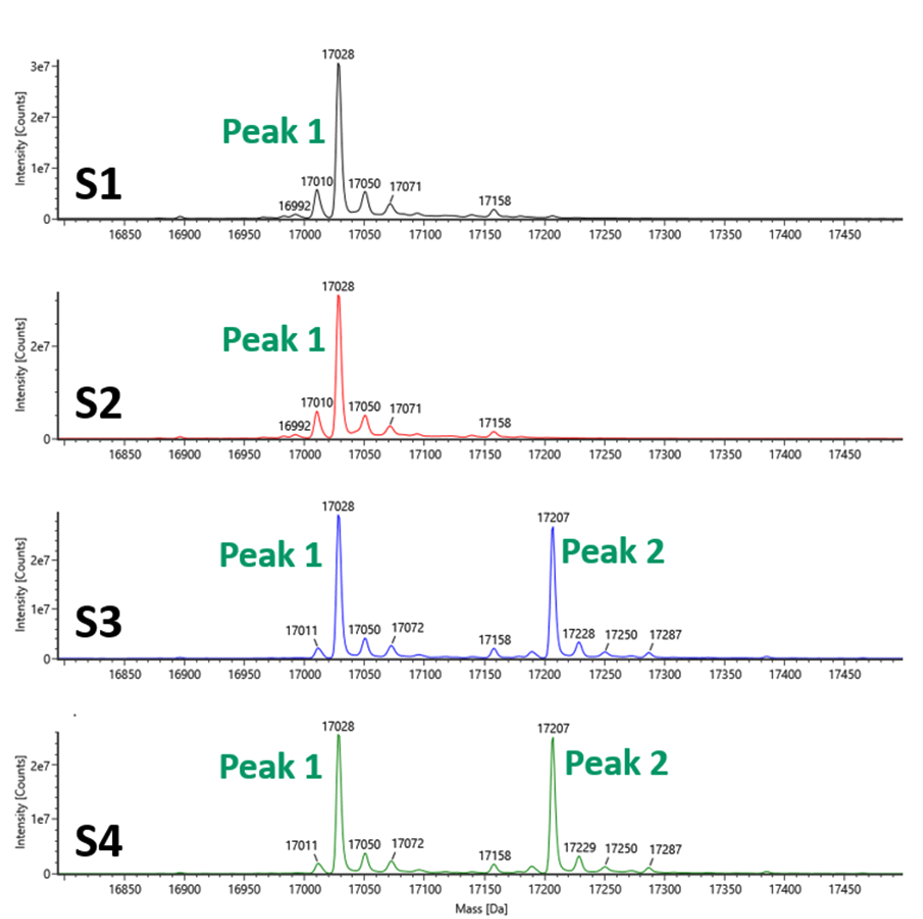
Figure 4. Variation in Sample Quality Detected by Intact Mass
Conclusion
Since its launch, Paveway has proved its mettle as an E. coli expression system, with more than 150 molecules expressed in this platform at industry-leading product titers. The core Paveway workflow enables selection of a high-producing strain based on product titer in about six weeks from program initiation.
This case study was engineered to highlight the ability of the Paveway PLUS enhanced workflow to detect post translational modifications in partially purified product. The propensity of BL21 strains to gluconoylation was already known. We chose this system specifically to demonstrate the added value of the enhanced Paveway workflow.
The enhanced workflow takes 11 weeks compared to six weeks for the standard titer-based selection workflow but provides much more actionable data by enabling strain selection based on both titer and product quality, protein characterization that includes to identification of product-related impurities, and last but not least from a manufacturing perspective, identification of a suitable resin for capture chromatography.
We previously identified bioreactor 5 as the top-producing strain by titer, but it turned out to be 50% gluconyolated. Had this set of conditions been taken forward without the advanced workflow, a client would have not discovered this post-translational modification in their product until very late in development.
The enhanced Paveway PLUS workflow identifies post-translational modification early, requiring only small quantities of partially purified sample, and suggests optimized expression systems, production methods, workflows, and quality analytics in just eleven weeks.
Learn more about Paveway™ PLUS
Authors:
Authors are located at FUJIFILM Diosynth Biotechnologies, UK and RTP, NC*
Chris Lennon Director of the Microbial Upstream Process Development Group
Jodie Symmington Staff Scientist (DSP HTP Automation)
Emma Jackson Principal Scientist within Process Development
Paul Stephenson Principal Scientist within Analytical Development
Jessica Kane-Fidgeon Principal Scientist within Analytical Development
Jeff Keen Senior Staff Scientist within Analytical Development
Rebecca Abram Director of the Strategic Technical Marketing Team
Steve Loftus Microbial Business Steering Group Lead
Francisco Gonzalez Associate Director of the Strategic Technical Marketing Team*
F[1] Stumpp MT, Amstutz P. DARPins: a true alternative to antibodies. Curr Opin Drug Discov Devel. 2007 Mar;10(2):153-9. PMID: 17436550.
Related information
Paveway™ PLUS Microbial Expression Platform
Our Microbial Service Offering